We are excited to announce that ZERAMEX®USA has been selected to become the front cover of the international C.E. magazine of oral implantology, Implant Tribune for their April 2020 issue!
Tag Archives: Zirconia Ceramic Dental Implants
Site #3: Patient requests removal of a titanium implant with peri-implant disease and replacement with a metal-free Zeramex XT implant; Case Presentation by Michael Toffler, DDS; New York, NY; September 2019
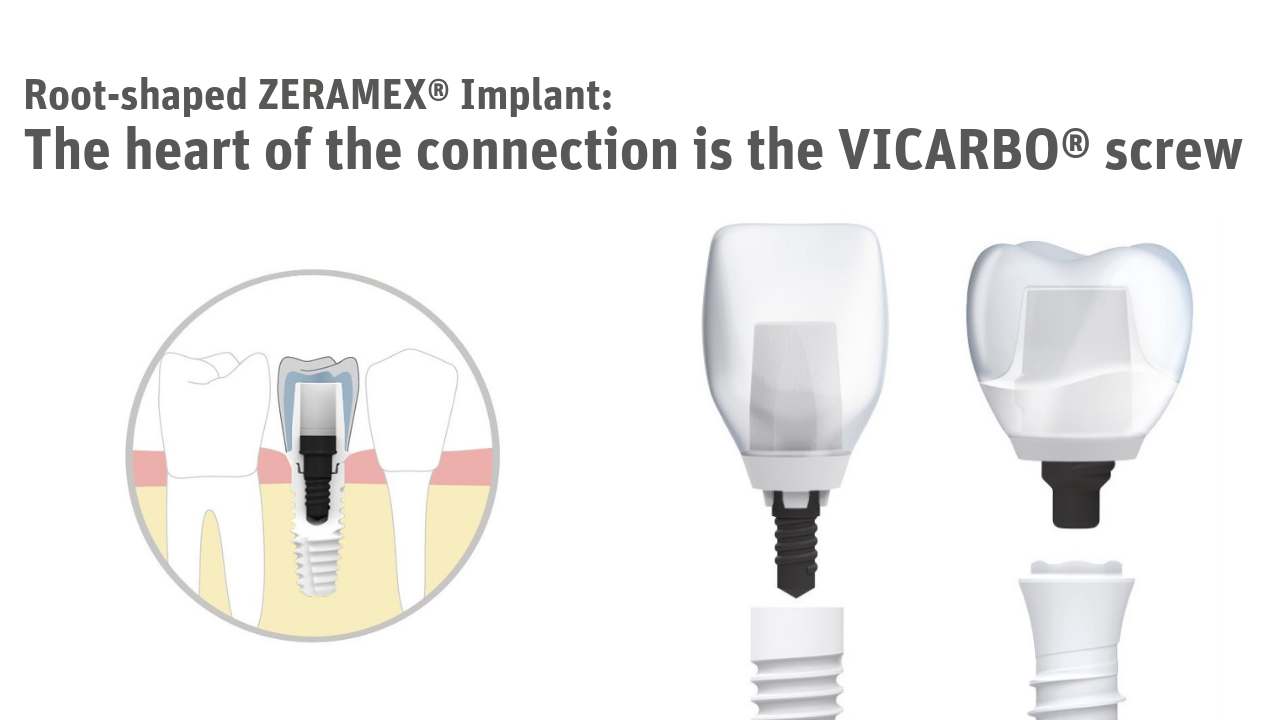
The main objective of ZERAMEX® was to offer a 100% metal-free solution in which not only the implant but also the screw are metal-free. The company therefore decided to use the high-performance material VICARBO® in their implant systems.
Dr. Peter S. Evans of The BioCompatible Difference interviews Michael Foley, Founder of Emerginnova – Zeramex USA and Dr. Sammy Noumbissi, President of the International Academy of Ceramic Implantology.
Dentalpoint with its ZERAMEX brand is a pioneer in “white implants”. In this interview with Ceramic Implants Magazine, Philip Bolleter, Head of the Technical Department, and Adrian Hunn, Head of Marketing and Sales, discuss the current state of ceramic implants.
Treatment planning and surgical placement of the FDA approved ceramic dental implant
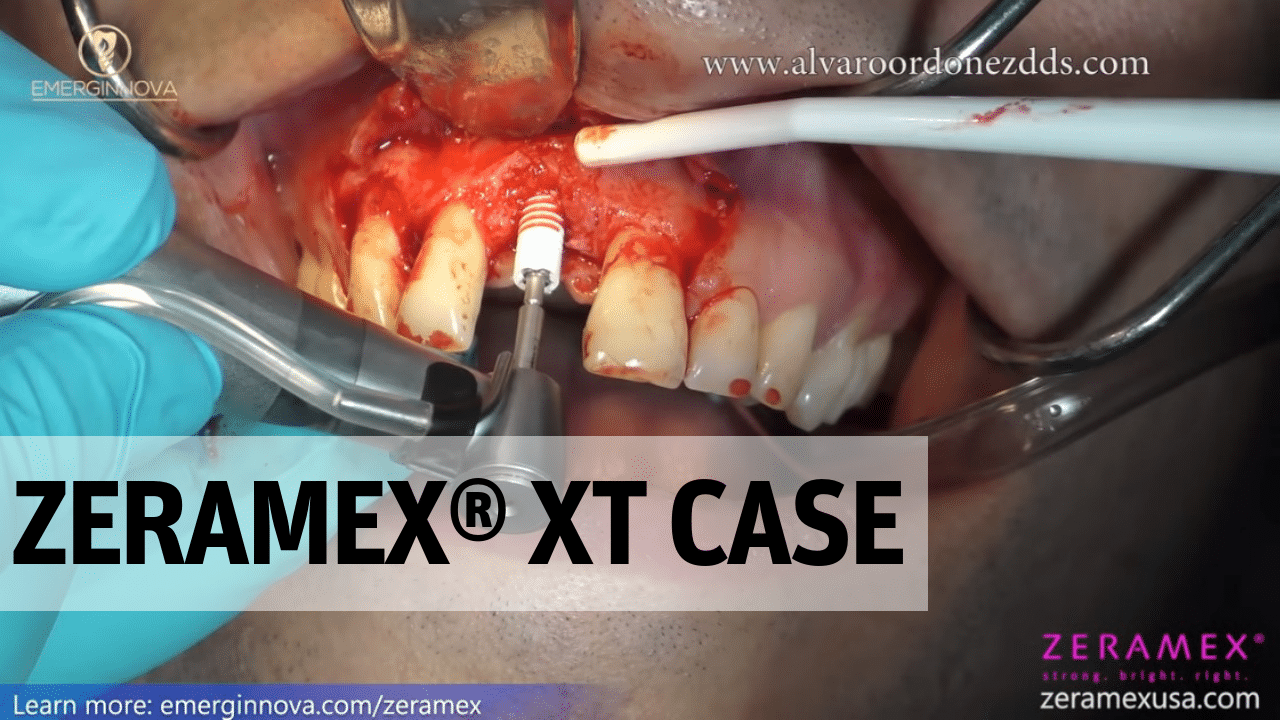
ZERAMEX® XT by Dr. Alvaro Ordonez.
The IABDM will be holding parallel meetings: one for dentists, doctors, and other health professionals, and one for the general public! The professional meeting kicks off on Thursday, November 14, when you can take one of several day-long courses for additional CEUs. The main meeting begins bright and early the following morning, as do multiple tracks of sessions for the general public.